Imagine a wound that refuses to heal despite months of treatment. The patient receives antibiotics, undergoes regular dressing changes, and follows all medical advice—yet the wound persists, trapped in a cycle of inflammation and infection. This frustrating scenario affects millions of patients worldwide, and science now understands why: biofilms.
Recent research estimates that biofilms are present in almost all chronic wounds, with studies reporting prevalence rates between 60% and 100% in non-healing wounds. These microscopic communities of bacteria create a nearly impenetrable fortress that renders conventional antimicrobial treatments largely ineffective. According to a 2025 study published in Biomedical Journal, global healthcare costs associated with biofilm-induced chronic infections are estimated at approximately $386 billion annually.
For healthcare professionals treating chronic wounds—whether diabetic foot ulcers, pressure injuries, or surgical site infections—understanding biofilms is no longer optional. It’s essential.
What Are Biofilms? Understanding the Architecture
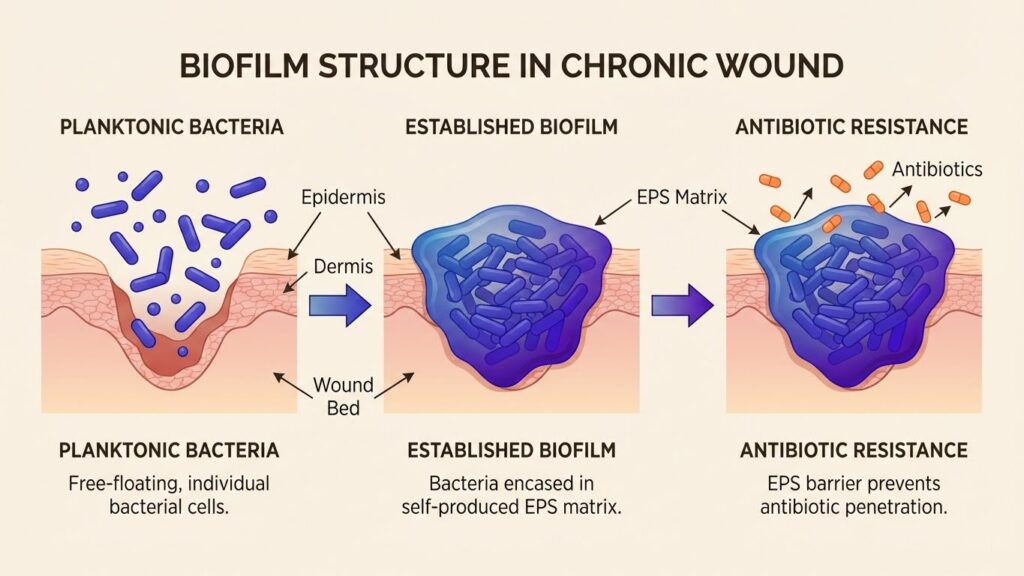
Biofilms are structured communities of bacterial cells that adhere to surfaces and encase themselves in a self-produced extracellular polymeric substance (EPS). Think of them as bacterial cities, complete with infrastructure, communication systems, and sophisticated defense mechanisms.
The EPS matrix consists of:
- Polysaccharides – providing structural support
- Proteins – facilitating adhesion and nutrient transport
- DNA – contributing to matrix stability
- Lipids – creating hydrophobic barriers
This complex architecture creates multiple layers of protection. The outer layers of bacteria may be metabolically active, while those deep within enter a dormant “persister” state that makes them extraordinarily resistant to antimicrobial agents.
The Biofilm Formation Process
Biofilm development follows a predictable sequence:
Stage 1: Initial Attachment Planktonic (free-floating) bacteria encounter a wound surface and begin adhering through weak, reversible bonds. This stage typically occurs within minutes to hours.
Stage 2: Irreversible Attachment Bacteria strengthen their attachment through specific adhesins and begin producing EPS components. This transition marks the point where simple cleansing becomes insufficient.
Stage 3: Maturation The bacterial community grows and differentiates. Microcolonies form, and the EPS matrix thickens. Channels develop within the biofilm, allowing nutrient distribution and waste removal.
Stage 4: Dispersion Bacterial cells detach from the mature biofilm and colonize new areas. This stage explains why biofilms can spread rapidly within wounds and why recurrence is so common.
Why Traditional Antibiotics Fail Against Biofilms
The therapeutic failure of conventional antimicrobials against biofilm-associated wound infections stems from multiple mechanisms:
1. Physical Barrier Protection The EPS matrix acts as a molecular sieve, slowing antibiotic penetration. By the time antimicrobial agents reach bacteria in the biofilm’s deeper layers, concentrations may be 100-1,000 times lower than necessary for bacterial killing.
2. Altered Microenvironment The biofilm interior creates unique conditions—low oxygen tension, acidic pH, and nutrient limitation—that fundamentally alter bacterial metabolism. Many antibiotics target actively growing bacteria, making them ineffective against slow-growing or dormant biofilm cells.
3. Persister Cells These are phenotypic variants that enter a dormant state, making them inherently tolerant to antibiotics. Research shows that persisters can represent 1-10% of biofilm populations. They survive antibiotic treatment and repopulate the biofilm once treatment ceases.
4. Increased Genetic Exchange The close proximity of bacterial cells within biofilms facilitates horizontal gene transfer, including antibiotic resistance genes. This creates hotspots for the evolution and spread of antimicrobial resistance.
5. Quorum Sensing Communication Bacteria within biofilms use chemical signaling molecules to coordinate behavior, including upregulation of resistance genes and production of enzymes that inactivate antibiotics.
The Clinical Impact: Why Biofilms Matter
The presence of biofilms fundamentally changes wound healing dynamics:
- Prolonged Inflammation: Biofilms sustain a chronic inflammatory state, preventing progression through normal healing stages
- Impaired Tissue Repair: Continuous bacterial presence disrupts cellular and molecular mechanisms essential for wound closure
- Increased Healthcare Burden: Patients with biofilm-infected wounds require longer hospital stays, more frequent interventions, and higher healthcare costs
- Amputation Risk: In diabetic foot ulcers, biofilm presence significantly increases the risk of limb loss
A 2025 consensus document from the World Union of Wound Healing Societies now states that “all non-healing chronic wounds potentially harbor biofilms” and recommends that treatment should target biofilm disruption and prevention of reformation.
Current Approaches to Biofilm Management
The medical community has developed a multi-faceted strategy known as Biofilm-Based Wound Care (BBWC):
Aggressive Debridement Mechanical removal of biofilm through sharp debridement, ultrasonic methods, or enzymatic approaches. This temporarily reduces bacterial burden and makes remaining bacteria more susceptible to antimicrobials.
Cleansing and Irrigation Use of wound cleansers to physically remove planktonic bacteria and debris. However, standard irrigation alone cannot eliminate established biofilms.
Antimicrobial Dressings Application of silver, iodine, or other antimicrobial-containing dressings. These can help suppress biofilm reformation but often cannot penetrate established biofilms.
The “Step-Down Approach” This strategy involves repeated debridement followed immediately by antimicrobial application, with treatment intensity adjusted based on healing progress.
The Challenge Remains: Need for Innovation
Despite these interventions, biofilm management remains frustratingly difficult. Current approaches face several limitations:
- Debridement provides only temporary relief; biofilms can reform within 24-48 hours
- Traditional antimicrobials struggle to penetrate the EPS matrix
- Resistance development continues to accelerate
- Treatment protocols lack standardization across clinical settings
- Cost and time burden on healthcare systems continues to grow
The Promise of Next-Generation Solutions
The biofilm challenge has driven innovation in antimicrobial development. Researchers are pursuing several promising approaches:
1. Biofilm-Penetrating Agents New antimicrobials designed to disrupt or penetrate the EPS matrix, reaching bacteria that conventional agents cannot touch.
2. Anti-Biofilm Enzymes Enzymes that specifically target components of the EPS matrix, breaking down the protective structure.
3. Quorum Sensing Inhibitors Molecules that disrupt bacterial communication, preventing coordinated biofilm formation and maintenance.
4. Combination Therapies Synergistic approaches that combine physical disruption with multiple antimicrobial mechanisms.
5. Advanced HOCl-Based Solutions Next-generation hypochlorous acid formulations that maintain efficacy in the presence of organic matter—a critical limitation that has prevented traditional HOCl products from addressing biofilms effectively.
Looking Forward: A New Era in Wound Care
The biofilm challenge represents one of the most significant obstacles in modern wound care. As our understanding of these bacterial communities deepens, so does our ability to develop targeted interventions.
Recent advances in antimicrobial chemistry, particularly formulations that maintain stability and efficacy in challenging wound environments, offer hope for improved outcomes. The key lies in solutions that can:
- Penetrate established biofilm matrices
- Remain effective at high dilutions with wound exudate
- Avoid promoting antimicrobial resistance
- Support, rather than impair, the wound healing process
For healthcare providers on the front lines of wound care, staying informed about biofilm biology and emerging treatment strategies is essential. The patients whose wounds refuse to heal deserve access to innovations that finally break through the biofilm barrier.
Conclusion
Biofilms represent a formidable challenge in chronic wound management, but they are not insurmountable. Through continued research, clinical innovation, and the development of next-generation antimicrobials specifically designed to overcome biofilm protection mechanisms, we are entering a new era in wound care.
The goal is clear: transform chronic, non-healing wounds into acute, healing wounds by effectively managing the biofilm communities that perpetuate infection and inflammation. With the right tools and approaches, this goal is increasingly within reach.
